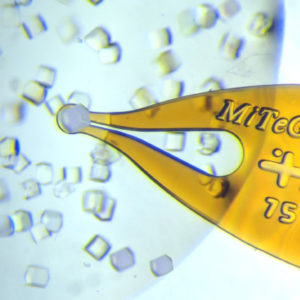
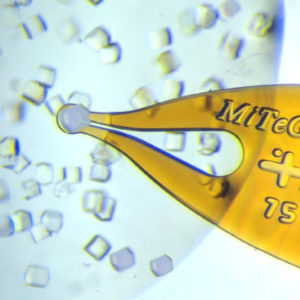
上海金畔生物代理MiTeGen品牌蛋白结晶试剂耗材工具等,我们将竭诚为您服务,欢迎访问MiTeGen官网或者咨询我们获取更多相关MiTeGen品牌产品信息。

LV CryoOil
- Superior Cryoprotectant
- Ultra-low viscosity
- Minimal background scatter
Product Information
- Product Description and Usage
- Why Mitegen’s LV CryoOil™
Oils are widely used in crystallography – as cryoprotectants when samples are flash cooled, and as barriers to the dehydration, solvent evaporation, and oxidation that may occur in air near and above room temperature.
The ice rings seen in the diffraction patterns of flash cooled protein crystals arise primarily from crystallization of the aqueous solution surrounding the crystal, not of the internal solvent. Oils can displace and replace this surrounding solution with little risk of damage due to osmotic shock.
Advances in X-ray sources, optics, detectors and sample mounting technology have allowed protein crystals as small as 5 microns to be used in structure determinations. Crystals smaller than roughly 50 microns dehydrate rapidly in air, with sub-10 micron crystals dehydrating in seconds.
Substantial dehydration-induced damage can easily occur between removal from a drop and flash cooling. Dehydration can be minimized by first transferring to an oil drop (using the MicroSieves™ in Microtools™ Kit 1 and Microtools™ Kit 2 ) before mounting the oil-coated crystal on a MicroMount or MicroMesh.
However, surrounding oil scatters X-rays, reducing diffraction signal-to-noise, and contributes thermal mass that reduces crystal cooling rates and increases the need for penetrating cryoprotectants. These problems are especially acute for the smallest crystals. When mineral oil, Paratone oil and other high viscosity oils are used, the volume of surrounding oil can exceed the crystal volume by a factor of 10 or more, and can contribute excess drag that increases sample motion in a cryostream. For larger rod or plate shaped crystals, the surrounding oil exerts stresses when it freezes that can cause crystal cracking and mosaic broadening.
You may also like…
-
MicroTools
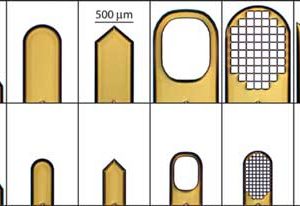 These tools have tips made from soft, flexible microfabricated polymer films. Tip curvature gives them…
These tools have tips made from soft, flexible microfabricated polymer films. Tip curvature gives them… -
Crystal Mounts and Loops
 Simplify crystal harvesting, and obtain the best possible data from your crystals, using the most advanced…
Simplify crystal harvesting, and obtain the best possible data from your crystals, using the most advanced…
MicroTools
Crystal Mounts and Loops